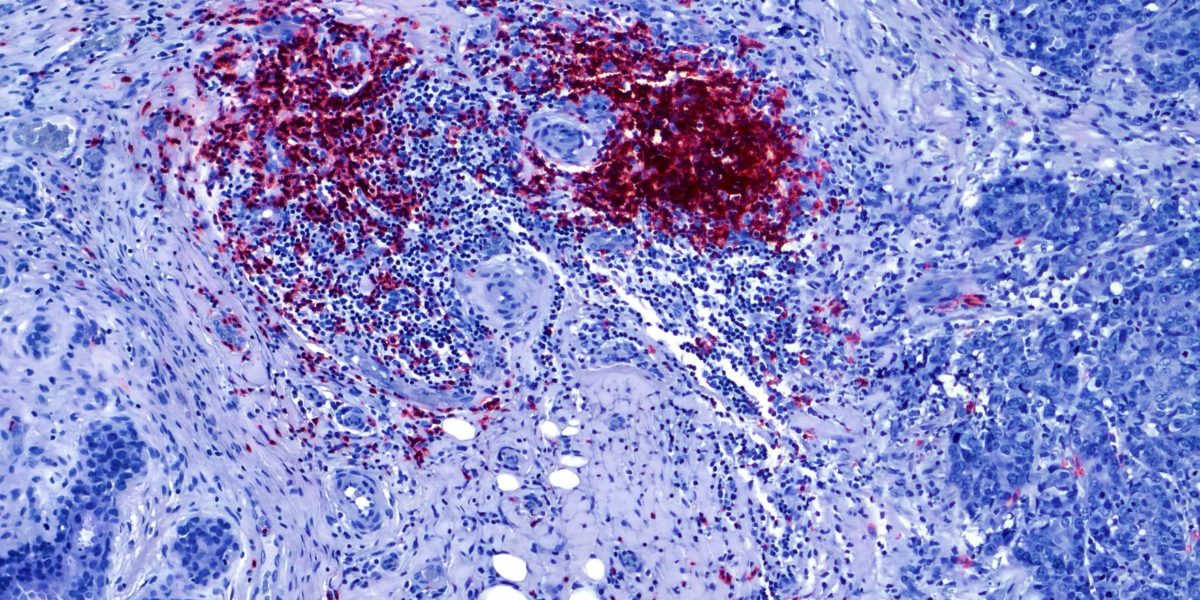
In breakthrough research at ChristianaCare’s Helen F. Graham Cancer Center & Research Institute, scientists have discovered that a protein secreted by tumor cells can switch off the body’s natural defenses against triple negative breast cancer (TNBC).
The study, led by Jennifer Sims-Mourtada, Ph.D., lead research scientist at the Cawley Center for Translational Cancer Research (CTCR), at the Graham Cancer Center, is reported in “The Journal of Translational Medicine” which is available online.

“What we found is that TNBC tumor cells can effectively shut down the body’s defense systems against the tumor by secreting a type of protein called IL-10,” Dr. Sims-Mourtada said.
“The presence of this immune system protein forces the antibodies that would normally be created to attack the tumor to become non-reactive and not do what they are supposed to do.”
The study was initiated in partnership with The Wistar Institute of Philadelphia, Pennsylvania, in collaboration with the late Raj “Shyam” Somasundaram, Ph.D., a cell biologist at the Melanoma Research Center.

“Dr. Sims-Mourtada and her team have brought us tantalizingly close to understanding what drives the aggressive nature of triple negative breast cancer, a treatment-starved disease that disproportionately affects Delaware women,” said Nicholas J. Petrelli, M.D., Bank of America endowed medical director of the Helen F. Graham Cancer Center & Research Institute.
“Their work underscores our belief that scientific collaborations such as this one between our Cawley CTCR clinicians and Wistar scientists can smooth the way for new findings to become effective therapies, especially for hard-to-treat and aggressive forms of cancer like TNBC.”
Understanding the mechanism behind TNBC
Delaware ranks highest in the nation for incidence of triple negative breast cancer. TNBC is an aggressive form that affects Black women at twice the rate of white women with poorer outcomes. Patients have higher rates of early recurrence than other breast cancer subtypes, particularly in the first five years after diagnosis. Currently there is no targeted therapy for TNBC.
“One of our missions within the Cawley CTCR is to understand the mechanisms behind TNBC and find a treatment for it,” Dr. Sims-Mourtada said. “Our study sheds new light on what is prompting the body’s immune response to the cancer cells and offers clues to potential new therapeutic targets.”
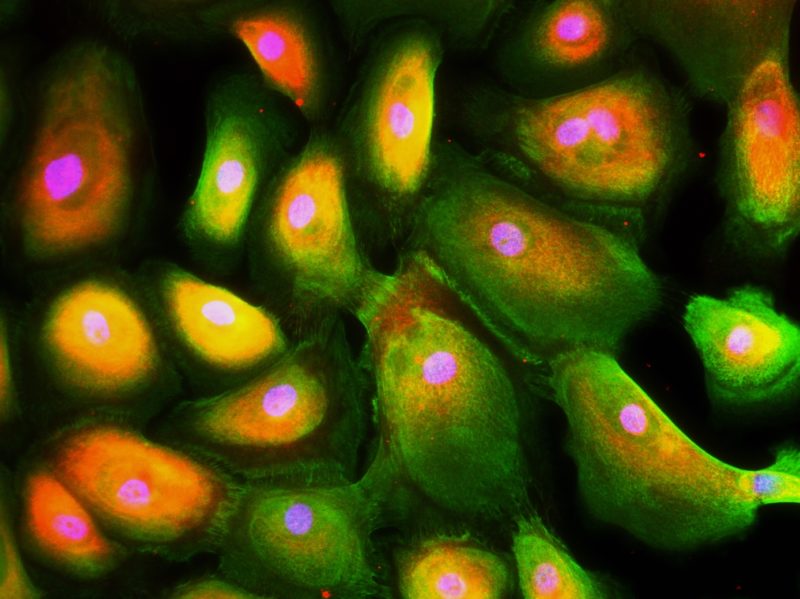
Normally it is the job of the B cells to regulate the immune response against foreign invaders like cancer. Among other jobs, they control inflammation at the site of an attack by releasing proteins, including IL-10, to signal the defender cells to stand down.
“Previously it was thought that the immune cells were the ones to express IL-10 to regulate themselves,” Dr. Sims-Mourtada said. “But our study shows that the tumor cells also release this protein, which means they are driving how the immune system behaves.”
Within the tumor microenvironment, IgG4 is one of four antibody subclasses expressed and secreted by B cells. Whereas another type of antibody would urge the immune system to press on with the attack, activation of IgG4 signals the job is done.
TNBC and activation of IgG4
“Our findings support that TNBC may create a tumor environment that supports activation of IgG4, and messaging from IL10 is triggering the switch,” Dr. Sims-Mourtada said.
As previously reported with other cancers, such as melanoma, this study confirms that the presence of IgG4-positive B cells within the tumor associates with advanced disease increased recurrence and poor overall breast cancer survival. It is also possible that IL-10 expression by tumor cells may also be a cause of poor outcomes in TNBC, and this may be independent of IgG4+ B cells.
“At this point, we don’t know what causes tumor cells to start secreting IL-10, but we know that B cell-tumor cell interactions are involved,” Dr. Sims-Mourtada said.
“We still have to look at what is really going on in the B cell population to determine which subtypes of B cells are affected by this tumor crosstalk and why some forms of TNBC express IL-10 (the ones with poor outcomes) and others do not.
“We think that the presence or absence of other immune cells in the microenvironment may affect how B cells interact with tumor cells to drive IL-10 expression,” she said.

Resources for the study, including blood and tissue samples from consenting patients, were obtained through the Graham Cancer Center’s Tissue Procurement program. Interestingly, in a small subset of samples, the researchers found that IL-10 expression was significantly higher in Black patients than non-Hispanic white patients. These findings need to be confirmed in a larger more diverse population with different TNBC subtypes.
Understanding tumor-infiltrating B cells
“Our growing understanding of the contribution of IgG4+ cells to the immune microenvironment of TNBC and what drives IL-10 expression may reveal ways in which tumor-infiltrating B cells can contribute to tumor growth and provide new targets to increase the immune response to TNBC,” Dr. Sims-Mourtada said.
As partners for more than a decade, Graham Cancer Center research clinicians and Wistar scientists collaborate across disciplines to translate cancer research into more effective therapies for patients everywhere. In addition to providing high-quality, viable tissue samples for Wistar research studies, Graham Cancer Center clinicians actively participate in concept development, sharing their unique understanding of the everyday patient experience.