A research team at Christiana Care Health System’s Helen F. Graham Cancer Center & Research Institute found that a new mammography approach, synthesized 2D digital breast tomosynthesis (DBT-s2D), had lower recall rates and less radiation exposure to patients, while maintaining a high standard for cancer detection.
The five-year study, “Clinical Performance of Synthesized Two-dimensional Mammography Combined with Tomosynthesis in a Large Screening Population,” published in the February issue of the peer-reviewed journal Radiology, compared the new DBT-s2D mammography approach to those used today, 2D and 3D – or full-field digital mammography alone and digital breast tomosynthesis with full-field digital mammography.

The results surprised and pleased the researchers, said Jacqueline S. Holt, M.D., FACR, director of Breast Imaging at the Helen F. Graham Cancer Center & Research Institute, senior author of the study.
“Not only did fewer women have to return for biopsies and additional testing with the new synthesized DBT approach, but a greater percentage of invasive cancers, which are the more lethal cancers, were found,” Dr. Holt said. “The cancer detection rate overall was similar between the synthesized approach and 3D, and the synthesized DBT approach exposed the women to less radiation.”
In the study, 78,810 mammograms were performed at the Breast Center between October 11, 2011, and June 30, 2016. Of those mammograms, 16,173 used the new synthesized method, 30,561 used 3D, and 32,076 used 2D.
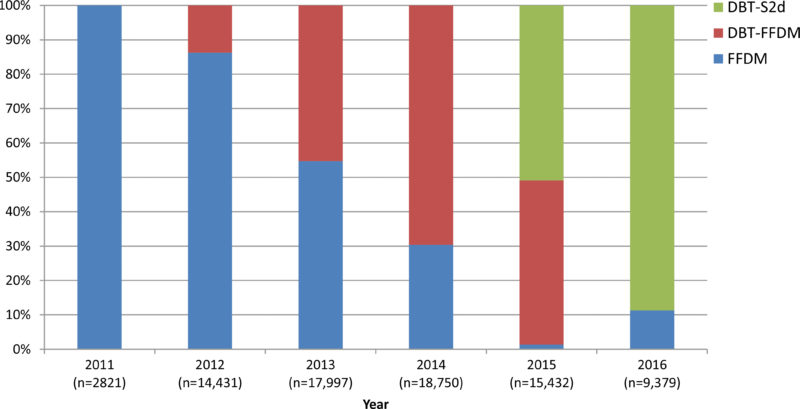
The call-back rate for the new synthesized method was 4.3 percent, compared to 5.8 percent for 3D and 8.7 percent for the 2D. One result that stood out was that the new synthesized method detected a statistically significant increase in the percentage of invasive cancers when compared to 3D – 76 percent compared to 61.3 percent.
“The significance of the results and what makes them so impressive is the number of mammograms in the study,” Dr. Holt said. “We could be seeing an important advancement globally in the quality of cancer care.”
“The entire staff at the Breast Center at the Graham Cancer Center is conscientious about optimizing patient care and adding value,” Dr. Holt said. “Lowering recall rates and biopsies is very important to us. Radiation exposure and additional testing are the so-called ‘harms’ of mammography, and we have them now down to the absolute lowest rate while continuing to maintain cancer detection rate.”
Dr. Holt said researchers at the Graham Cancer Center had expected the study would show the new synthesized mammography method to be equivalent to 3D, rather than to outperform it.
“Our radiologists at Christiana Care have embraced the new approach after seeing the results of this study,” Dr. Holt said. “The proof is in the pudding.”
The U.S. Food and Drug Administration (FDA) approved the 3D digital breast tomosynthesis with full-field digital mammography (DBT-FFDM) approach in 2011 after multiple research studies showed a reduction in the rate of call-backs, reduction in false positives and an increase in detection rate when compared to 2D full-field digital mammography (FFDM), the existing technology. The drawback with the DBT-FFDM approach is that it requires a second radiation exposure to the breast. Although this dual exposure does not exceed recommendations by American College of Radiology Mammography Quality and Standards, it is still 39 to 45 percent above that of FFDM. It also requires the patient to undergo increased time in breast compression.
Diagnostic imaging manufacturers began to develop new software techniques capable of producing a synthesized 2D image from 3D DBT data. In 2013, the FDA approved a version of synthesized 2D software (C-View, Hologic) designed to be used as an adjunct to DBT for breast cancer screening rather than FFDM.
Researchers at Christiana Care decided to study the new technology.

“With our focus of providing our patients with the best care available, we adopt the latest technologies if they prove themselves,” Dr. Holt said, predicting DBT-s2D will become a standard of care for those cancer centers with the technology because of the many clinical and cost benefits it brings. “I think we have shown at the Graham Cancer Center that we have decreased the risks in our mammography as much as possible and also have improved our overall clinical performance. The bottom line is we have proven that we provide a higher quality of care for our patients.”
Christiana Care’s Breast Center is a model for care across the country. It is the only facility in the region devoted exclusively to breast care, diagnosis and treatment. The Breast Center is available to all women who need routine screenings or further diagnostic procedures and treatments. Specialized services include the only dedicated breast MRI unit in the region, breast ultrasound, minimally invasive breast biopsies and clinical breast exams. Images are read on-site at the Breast Center, surgeons are available for consultation, and urgent cases are evaluated within 24 hours.
Thanks to a generous gift from philanthropist and businesswoman Tatiana Copeland, the Breast Center installed two 3-D mammography units in 2016.
In December 2016, the Breast Center earned a 2016 Guardian of Excellence Award from Press Ganey for patient experience in outpatient services.
The Graham Cancer Center is a national leader in research clinical trials and forms part of a select group of 51 research centers in the United States and Puerto Rico funded by the National Cancer Institute to participate in the NCI Community Oncology Research Program (NCORP). It also ranks 23rd among the 124 sites across the United States and Canada of the Alliance for Clinical Trials in Oncology for enrolling patients into trials.
Dr. Holt is grateful to Christiana Care for recognizing the value in pursuing the latest advances and technologies in cancer research and in supporting her and her co-authors Mireille P. Aujero, M.D., Sara C. Gavenonis, M.D., Ron Benjamin, D.O., and Zugui Zhang, Ph.D., as well as the entire research team and Christiana Care’s Value Institute for its contributions and statistical analysis.