Christiana Care pioneers U.S. trial of absorbable scaffold for heart patients
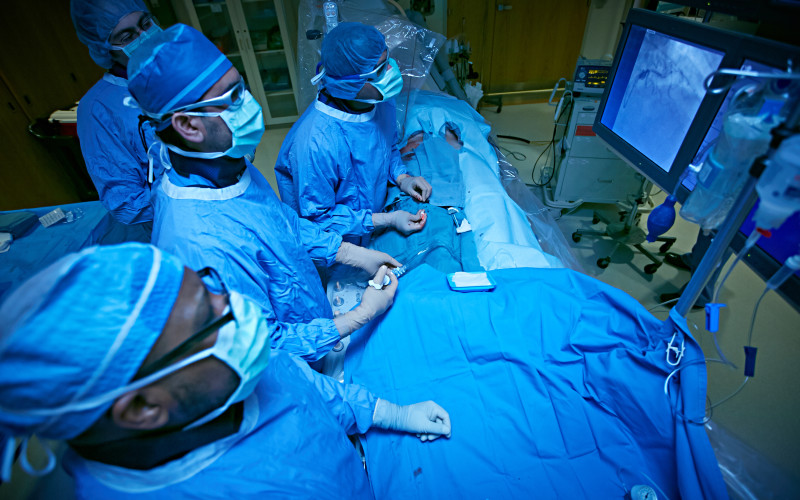
Christiana Care Health System is one of a handful of institutions in the region to participate in ABSORB III, the first clinical trial in the United States of an absorbable vascular scaffold for patients with coronary artery disease.
Unlike traditional metallic heart stents, the Absorb Bioresorbable Vascular Scaffold device made by Abbott opens blocked arteries with a mesh tube made of polylactide, a naturally dissolvable material commonly used in dissolving sutures. After the device is implanted the body absorbs it, allowing the artery the potential to regain its normal physiology.
“This could be a life-changing treatment for patients with coronary artery disease,” said Wasif Qureshi, M.D., medical director, Structural Heart Program, at Christiana Care’s Center for Heart & Vascular Health and the principal investigator. “We are always looking for innovative treatments that can lead to better outcomes and improve the lives of our patients.”
Heart disease is the leading cause of death for both men and women in the United States. Coronary artery disease is the most common type of heart disease and occurs when the arteries that supply blood to the heart become narrowed by a buildup of plaque, which is composed of fat, cholesterol, calcium and other deposits.
Since the 1970s, metallic stents have been used to open blocked arteries and deliver medicine to the vessels. The stent remained in the patient for the rest of his or her life. The Absorb device is called a scaffold because it is designed to be temporary.
After the scaffold is absorbed, it no longer takes up room in the artery, allowing greater blood flow. The artery also regains its ability to contract and expand naturally.
The Absorb device was approved in Europe in 2011, where an earlier trial determined that no patients developed clots within three years of receiving the scaffold. Absorb also is in use in the Middle East and parts of Latin American and the Asian Pacific. In the ABSORB III trial, the goal is to enroll 2,250 patients, mostly in the U.S., including about 20 at Christiana Care.
In the trial, two out of three patients will receive the scaffold, while one will receive a traditional metallic stent. Investigators will compare outcomes for the two groups, measuring the safety and effectiveness of the device.